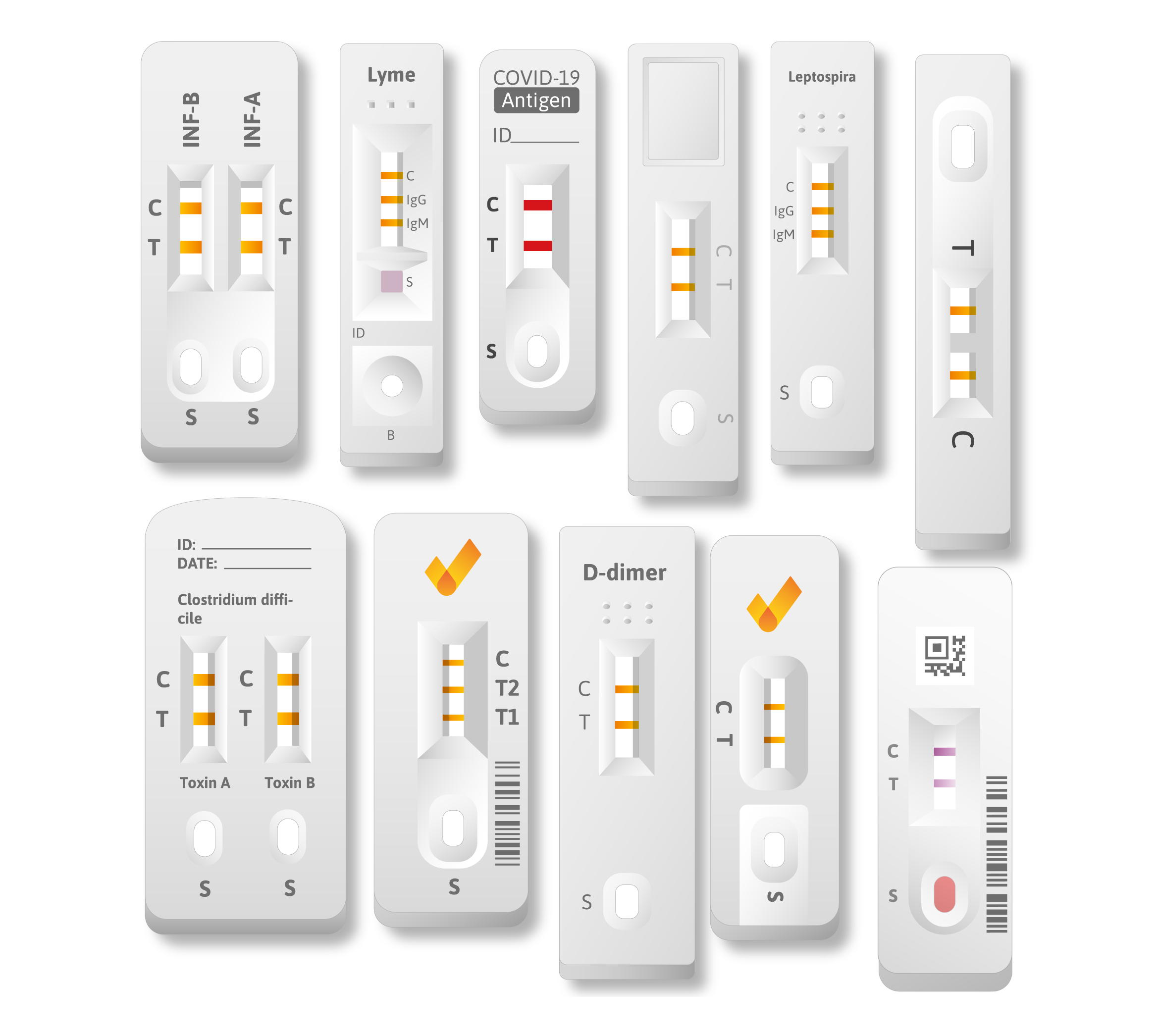
For several diagnostic applications, whenever time-to-result or the point-of-care is crucial, a rapid test can be used. There exist different types of rapid tests but focusing on human medicine, the ‘de facto’ standard is the so-called lateral flow assay. This assay can be analysed using photometric methods in order to obtain a real quantitative result.
A typical readout device is what we call a hardware reader. Hardware implies that we are talking about a real device that costs money, is an extra piece one has to carry around, has to be potentially updated and in some circumstances, is disposable.
As we can infer from the term photometric, we need a camera as the main ingredient of the overall reader recipe. This box can be surely ticked by a smartphone. Hence the raising power of ubiquitous smartphones and the increasing demand for telemedicine has forged the idea of using the device right at your fingertips to evaluate rapid tests, on-demand and on-premise.